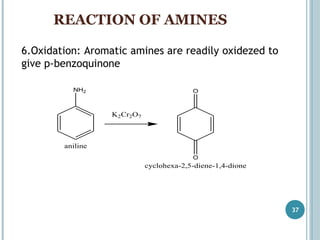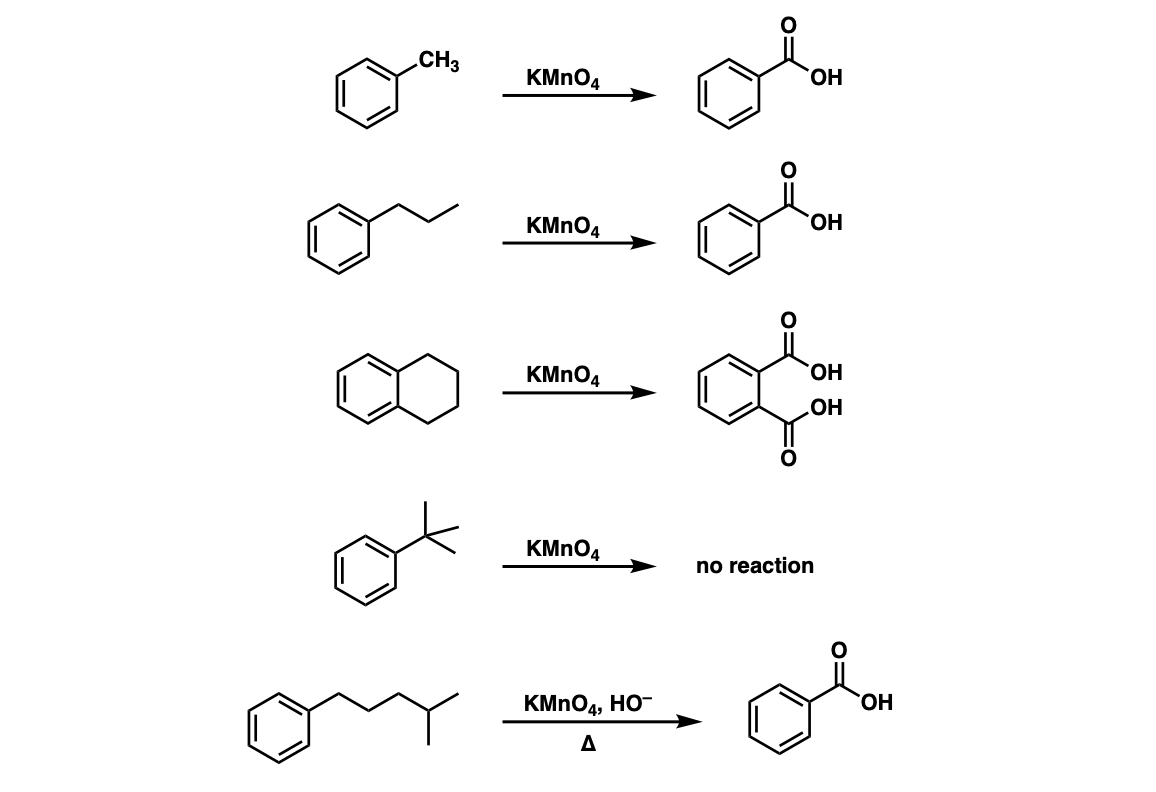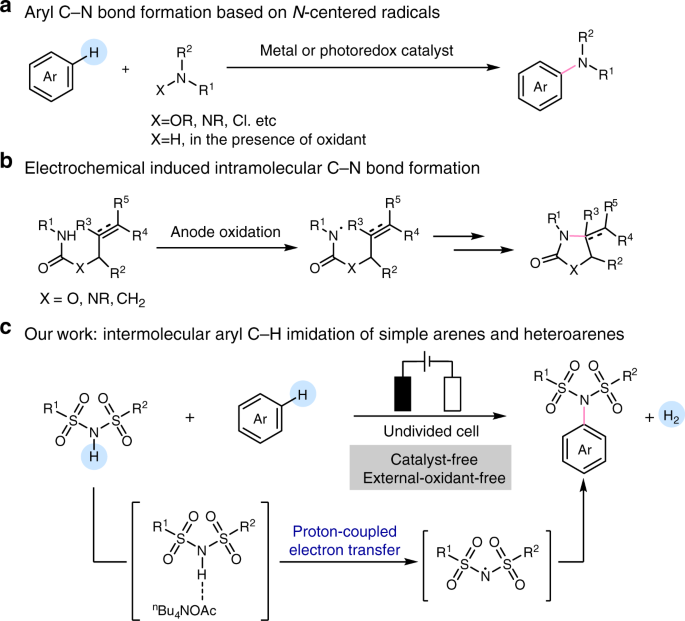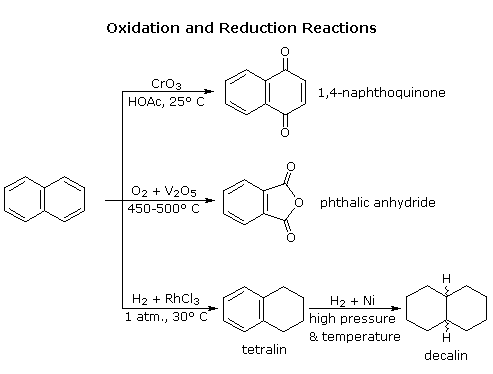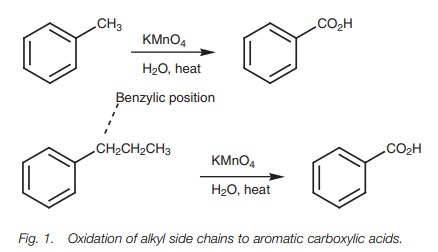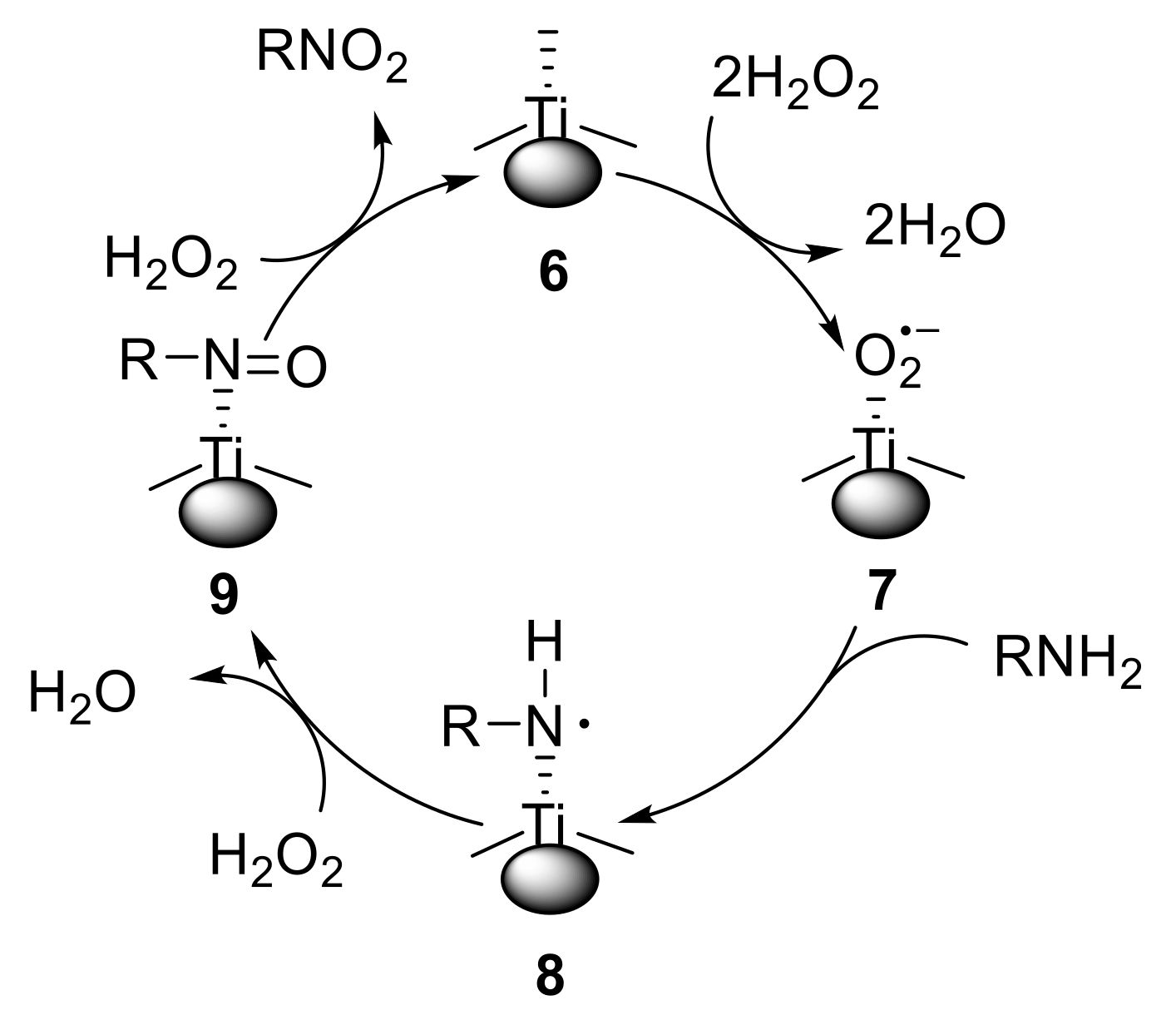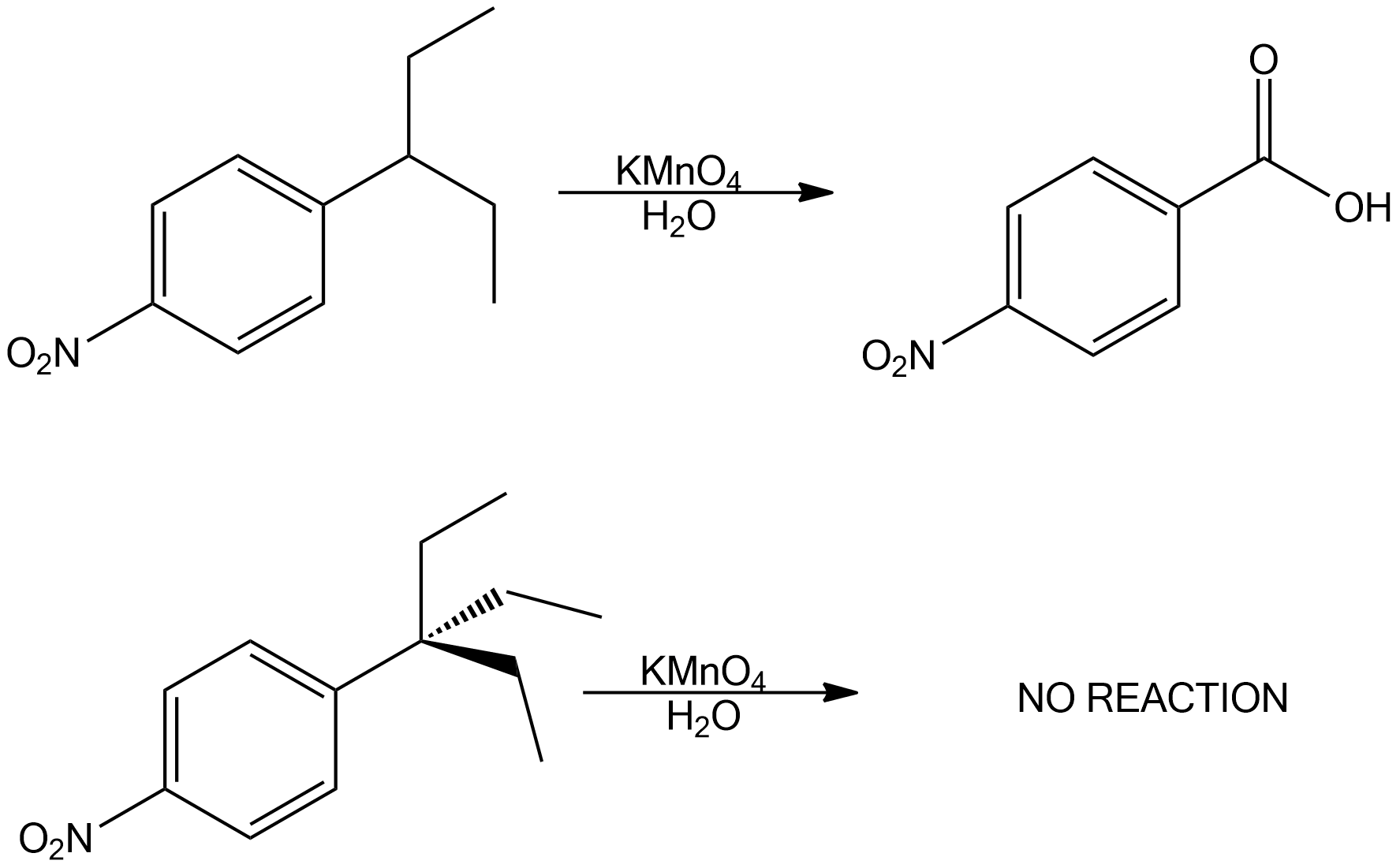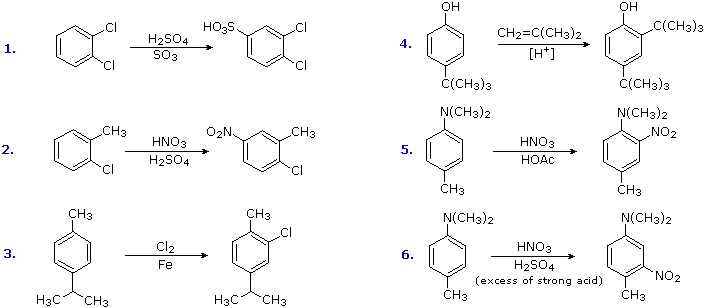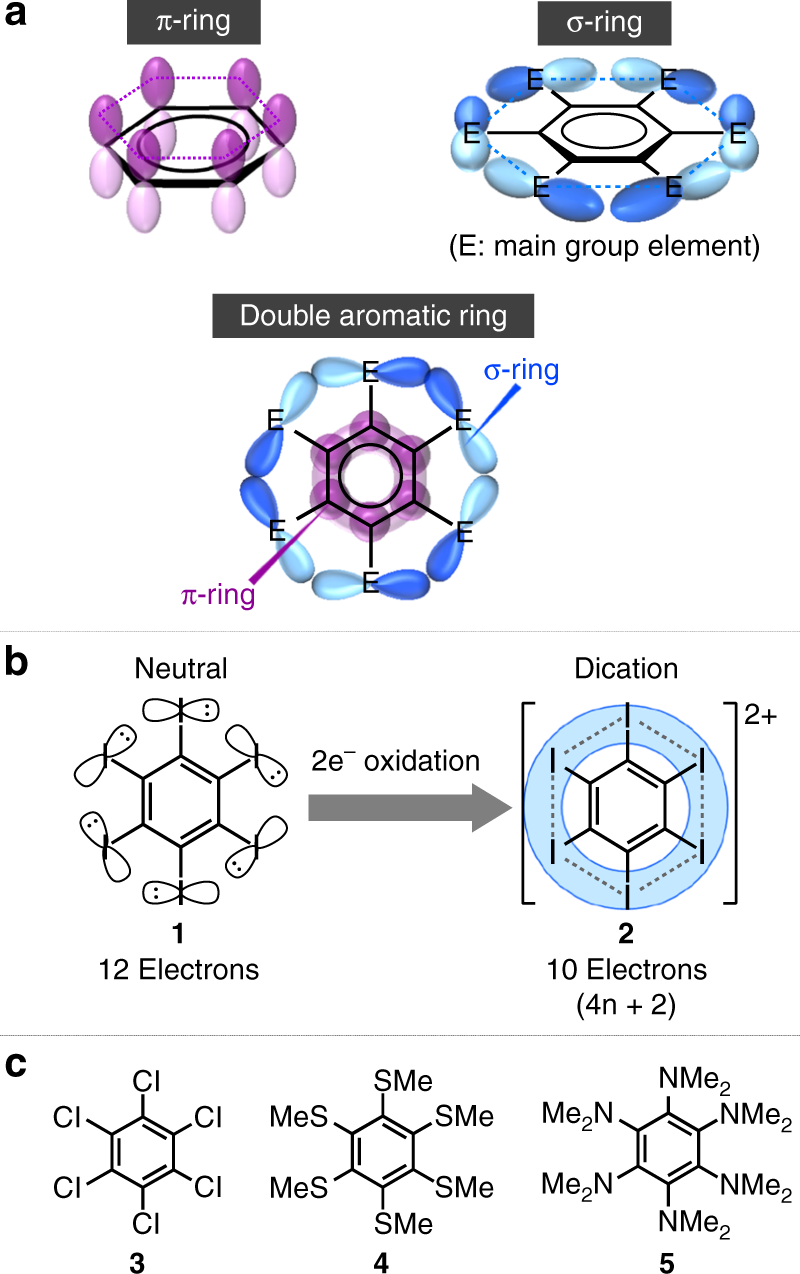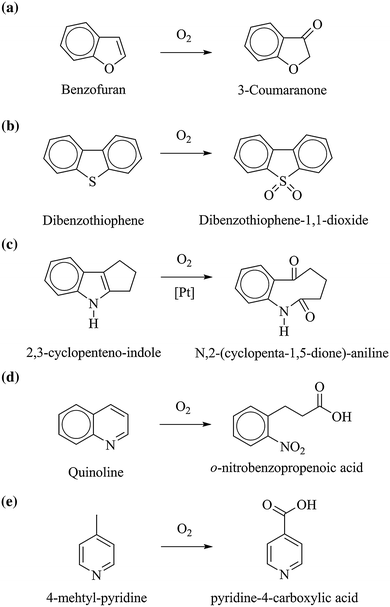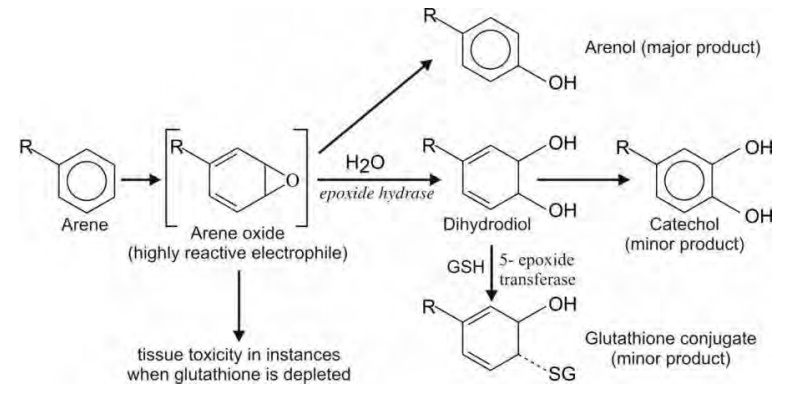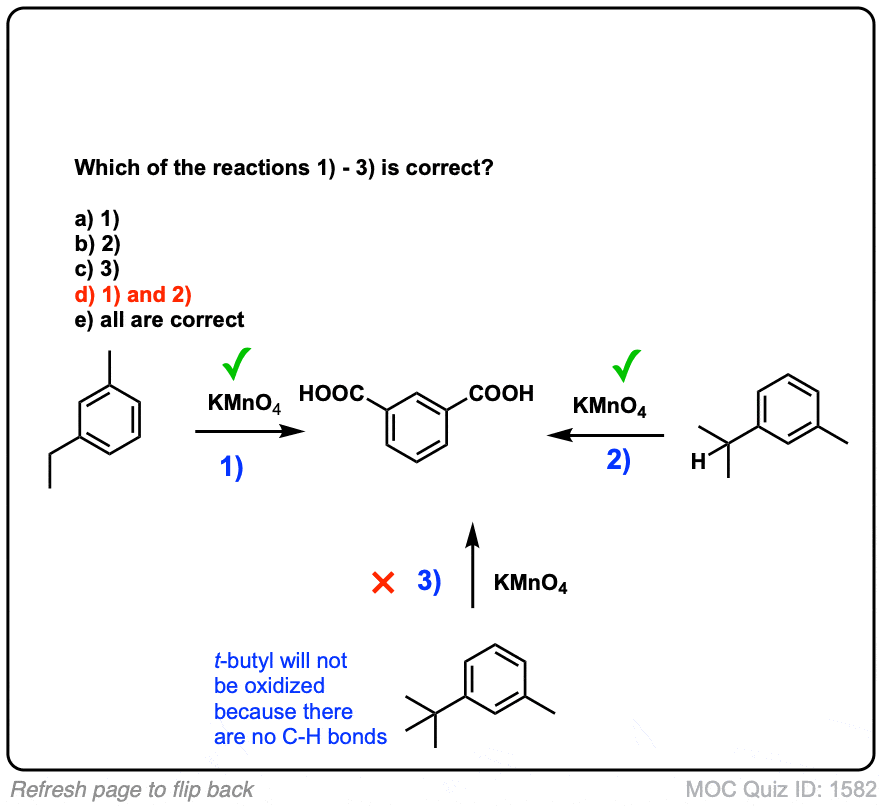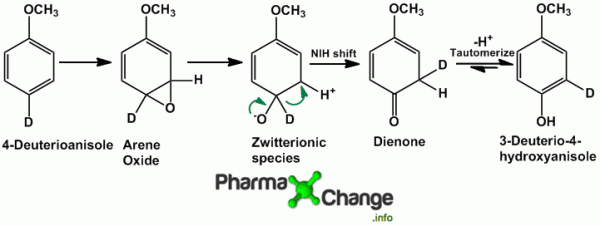
Oxidation of substituted aromatic hydrocarbons in the tropospheric aqueous phase: kinetic mechanism development and modelling - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C7CP08576A

Engineering Cytochrome P450 BM-3 for Oxidation of Polycyclic Aromatic Hydrocarbons | Applied and Environmental Microbiology

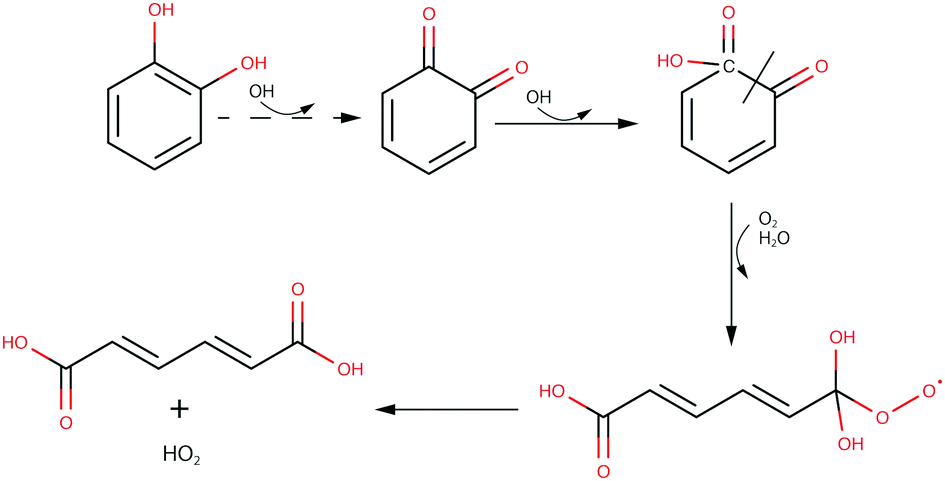
Oxidation Mechanism of Aromatic Peroxy and Bicyclic Radicals from OH−Toluene Reactions | Journal of the American Chemical Society
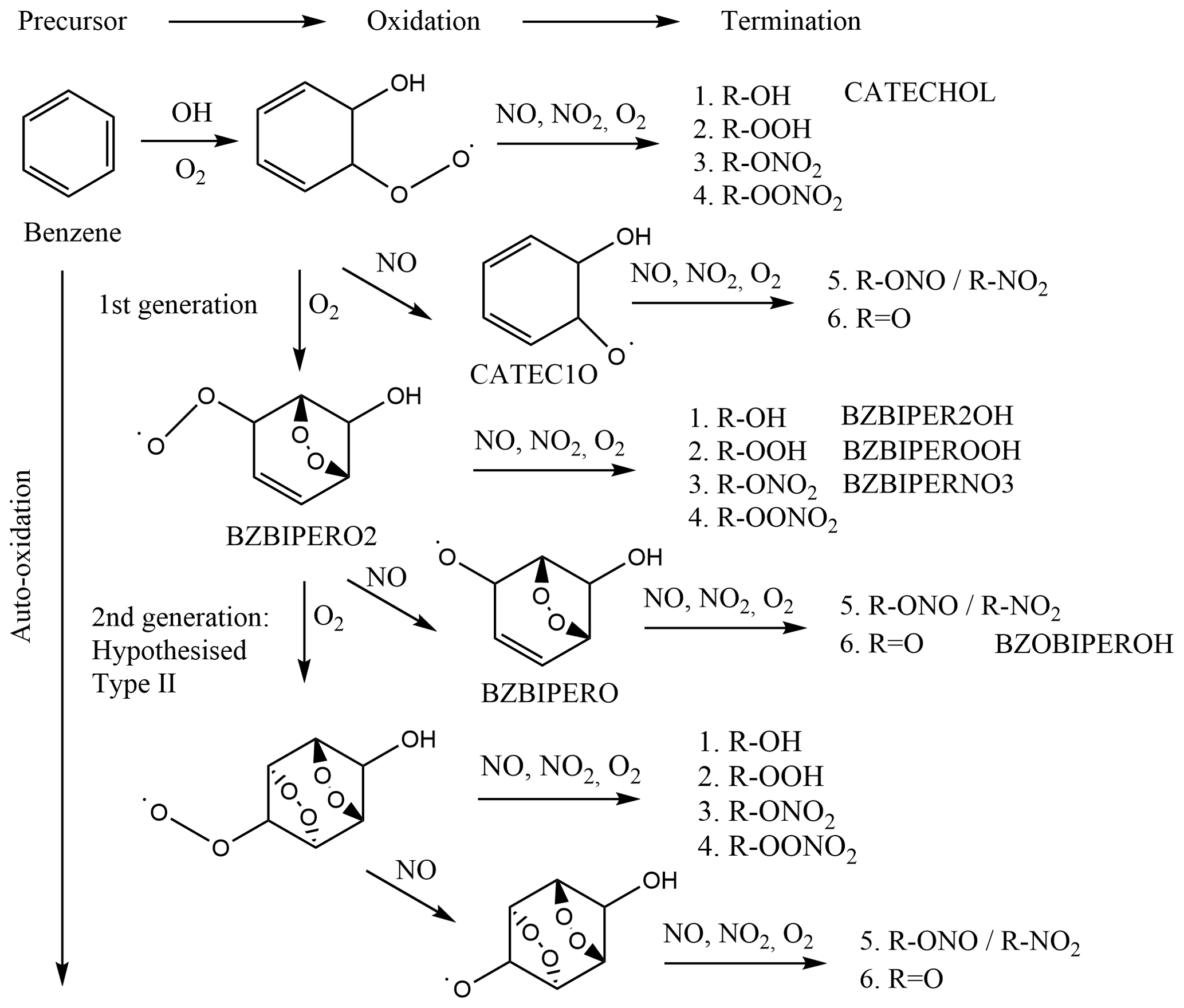
ACP - Chemical characterisation of benzene oxidation products under high- and low-NOx conditions using chemical ionisation mass spectrometry